Why should clinicians care about permittivity (ε) and conductivity (σ) when investigating breast cancer? Because these properties describe how tissues interact with microwaves, and they set the fundamental contrast that imaging systems and algorithms can exploit. In the breast, adipose tissue (low in water) typically shows lower ε and σ, while fibroglandular tissue is higher in both due to its water and ionic content. Many malignant lesions trend higher still. Our research project in this domain focuses on turning these physical differences into actionable indicators for screening, triage, and therapy monitoring.
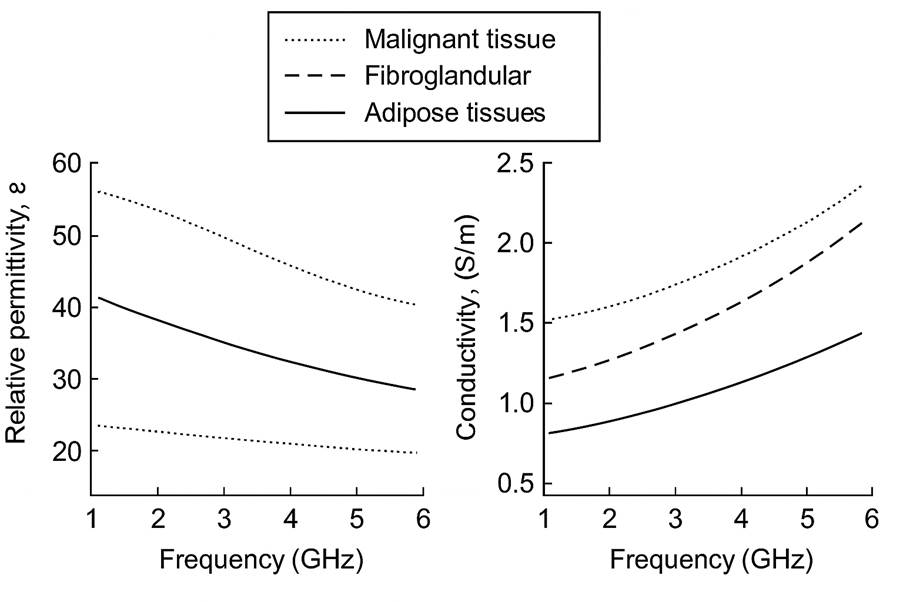
Where does dielectric contrast come from?
The origin of dielectric contrast is straightforward: water and ions drive the response. Decades of measurements show that tissues with more water content exhibit higher real permittivity and effective conductivity, and that both parameters vary with frequency. In the 1–6 GHz band often used for microwave breast imaging, this separation between fat and water‑rich tissues remains pronounced. Established references confirm this behavior. Gabriel et al.’s tissue catalogs consistently show higher ε and σ for water‑rich tissues compared to adipose, with characteristic dispersion across frequency. Large‑scale human studies such as Lazebnik et al. reported higher permittivity and conductivity in malignant breast tissues compared with normal and benign samples over an ultrawide bandwidth. The fundamental clinical question – “can we see anything relevant?” – directly depends on differences in ε and σ between regions. Together, these sources underpin a basic clinical truth: without physical contrast in ε and σ, there is no reliable image contrast to detect.
Translating physics into clinical tasks
Turning physics into clinically useful tasks begins with detection in screening and triage contexts. Systems must be sensitive to localized changes in ε/σ on length scales that matter, roughly 5–20 mm, which aligns with lesion sizes of interest for early intervention. That sensitivity depends on hardware configuration, coupling, calibration, and bandwidth, and it should be quantified transparently on well‑documented phantoms whose compositions reproduce realistic dielectric values over frequency. Beyond detection, triage benefits from characterization and diagnostic orientation: location, spatial extent, and possibly frequency‑dependent signatures that can improve specificity when combined with other modalities. For therapy monitoring, the emphasis shifts to temporal change, measuring relative variations in ε/σ that correlate with necrosis, edema, or tumor reduction over days to weeks, while controlling for temperature and hydration, which modulate dielectric properties.
Turning ε/σ into actionable “parameters”
For clinical communication, raw ε and σ cannot be presented directly. Instead, they inform derived indicators that answer practical questions. For example, a contrast map aggregates multi‑frequency information to highlight regions that deviate from a learned or measured background. A detection index could summarize the probability that a voxel or region exceeds a clinically tuned threshold, calibrated against phantom and retrospective data. Size estimates translate spatial contrast into an effective diameter with confidence bounds, acknowledging resolution limits and point‑spread effects. For longitudinal care, temporal stability metrics track contrast changes after correcting for instrument drift and thermal variation. These abstractions preserve the physics while speaking the language of “Is there a lesion? Where is it? How big is it? Is it changing?”
Practical constraints that shape clinical value
Several practical constraints shape how faithfully physics becomes clinical value. Temperature matters: both permittivity and conductivity shift with a few degrees Celsius, so measurements should be standardized or corrected. Anatomical variability matters, too: breast composition spans a continuum from adipose‑dominant to dense fibroglandular tissue, which influences background dielectric context and thus detection thresholds. Geometry and coupling influence near‑surface sensitivity; the presence of a skin‑mimicking layer and the properties of any supporting cup or tank alter fields and must be documented and compensated. Finally, frequency selection is not neutral: combining multiple bands can improve robustness when local uncertainties or artifacts impair single‑band performance.
How to validate translation to clinical practice?
Validation is the bridge from lab measurements to patient impact.
- Documented phantoms: A first stage relies on phantoms with clearly documented compositions and independently measured dielectric curves over different frequency bandwidths. Reporting size–contrast detectability on how small an inclusion can be reliably detected at given ε/σ contrasts helps set realistic expectations.
- Limited retrospective tests and feasibility: A second stage explores feasibility in limited, well‑controlled retrospective or prospective settings, emphasizing repeatability across days and temperature control.
- Pilot clinical studies: Pilot clinical studies then define appropriate endpoints on added sensitivity or positive predictive value in triage, localization error, size error, and temporal consistency, and examine how microwave‑derived indicators complement established modalities. This staged path keeps the physics honest while converging on clinical utility.
Our research follows this logic: start from well‑established dielectric behavior, design indicators that retain physical meaning yet are clinically interpretable and evaluate them progressively from standardized phantoms to carefully designed clinical pilots. Throughout, transparency about compositions, frequencies, measurement setups, and analysis methods is essential for reproducibility and for comparing results across groups.
Sources and further reading
- Gabriel, S.; Lau, R. W.; Gabriel, C. The dielectric properties of biological tissues: I–III. Phys. Med. Biol., 1996. https://doi.org/10.1088/0031-9155/41/11/001 | https://doi.org/10.1088/0031-9155/41/11/002 | https://doi.org/10.1088/0031-9155/41/11/003
- Lazebnik, M. et al. A large-scale study of the ultrawideband microwave dielectric properties of normal, benign and malignant breast tissues obtained from cancer surgeries. Phys. Med. Biol., 2007. https://doi.org/10.1088/0031-9155/52/20/002
- IT’IS Foundation Tissue Properties Database (frequency‑dependent tissue permittivity/conductivity and more). https://itis.swiss/virtual-population/tissue-properties/database/

 English | EN
English | EN 